Engineering the future of MedTech
Across Europe, engineers are designing a new generation of medical devices that are smaller, smarter and more practical than ever before. We are already seeing rapid progress on how we monitor health. In addition to large-scale machines in hospitals, engineers are developing ultra-thin ‘electronic skins’: flexible patches worn on the body that can track temperature, motion, hydration or sweat composition in real time. As Europe faces increasing pressure on healthcare systems, these innovations can help shape the future of how health is delivered. A recent review of on-skin flexible sensors for health monitoring shows how demanding this field can be. To work safely and comfortably, these devices must stretch and bend, breathe, adhere gently without irritation, protect delicate electronics from sweat, and still give precise readings. The authors highlight how silicones are key to enabling these technologies.
These are the kinds of breakthroughs that enable the innovation that is required to allow our healthcare systems to thrive in the future. The Horizon Europe work programme for 2026-2027, the New European Innovation Agenda and dedicated biotech and MedTech initiatives all aim to keep the EU at the forefront of medical technology, while strengthening strategic autonomy in critical health technologies and building resilient value chains.
A design toolbox for biomedical engineers
For biomedical engineers, silicones are not just ‘another material’, they can be viewed as a design toolbox. Silicone elastomers can be formulated from soft gels to durable rubber, combining excellent flexibility and fatigue resistance so that patches and catheters can bend thousands of times without cracking. At the same time, their chemical and thermal stability enable them to withstand repeated sterilisation, their electrical properties enable function, and their specific biocompatible and inert qualities protect patients’ tissues.
These properties give designers the freedom to create complex shapes and miniaturised components that combine soft tissue and hard electronics. Silicones help turn laboratory ideas into high-tech devices that can be manufactured at scale.
Enabling next-generation biomedical technologies
Silicones are central to the next wave of MedTech. A study on soft robotic micromachines for medical devices and personalised medicine explains how soft elastomers, such as silicones, are enabling soft robotic sleeves that gently support a failing heart, miniaturised, flexible tools for minimally invasive surgery, wearable and implantable systems that move and adapt to the body.
Over the lifetime of the device, this material stability helps maintain performance and supports compliance with technical standards on biocompatibility, electrical safety and hermetic sealing. Without materials that can be used in such versatile applications, many of these soft robotic ideas would remain on the drawing board.
Silicones, enablers of modern biomedical engineering
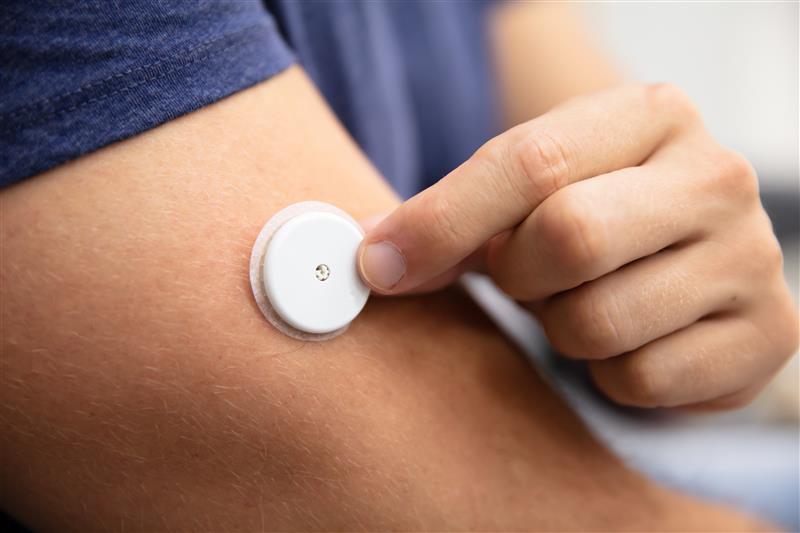
The study ‘Diabetes Technology Trends: a Review of the Latest Innovations’, provides another example of silicones supporting MedTech progress. Early glucose monitoring systems were large, hospital-based devices. Today’s continuous glucose monitors are roughly the size of a few stacked coins, worn for 7-15 days and sending data wirelessly to insulin pumps and smartphone apps. Silicones help these devices stay comfortably on the skin, ease the application, protect sensors and electronics from movement and body fluids, and keep performance stable over the full wear period.
To keep Europe at the forefront of biomedical engineering, policymakers must ensure that chemical and product regulations remain innovation-compatible, that research and innovation in advanced, biocompatible materials are supported, and that dialogue with the highly complex MedTech value chains is maintained. Protecting access to high-performance silicones is not just an industrial issue, it is a prerequisite for designing, building and scaling the next generation of medical technologies that can be made and used in the European Union.